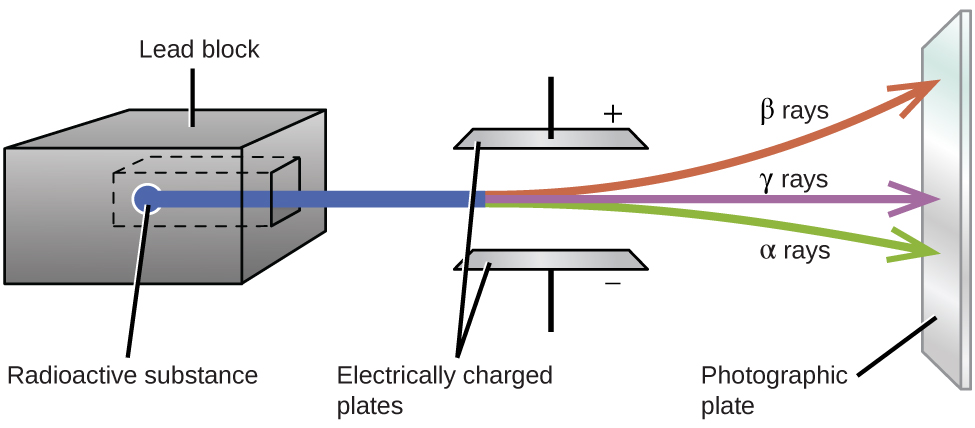
RADIOTOXINS¶
One of the big tasks facing humanity in the 21st Century is protecting itself from past screw-ups.
Humans destroyed two of their major cities, and spread radiotoxins around the planet as soon as they discovered how to make nuclear bombs (so-called "atom bombs").
Welcome to Planet of the Apes. "We are DEVO".
Before diving into atomic isotopes however, you might want to review the particle zoo.
Be sure to visit: Museum of Radiation and Radioactivity (ORAU)
Major Nuclear Accidents¶
- Three Mile Island
- Chernobyl
- Fukushima
%%HTML
<iframe src="https://giphy.com/embed/bSdkuLDnrYheE" width="480" height="196"
frameBorder="0" class="giphy-embed" allowFullScreen></iframe><p>
<a href="https://giphy.com/gifs/planet-of-the-apes-bSdkuLDnrYheE">via GIPHY</a></p>
from IPython.display import YouTubeVideo
YouTubeVideo("hRguZr0xCOc") # https://youtu.be/hRguZr0xCOc
from pandas import Series, DataFrame
# in years
half_life_table = {
'Pu238': 87.74,
'Pu239': 24_100,
'Pu240': 6500,
'Pu241': 14,
'I131' : 8.02/365.2181, # days per year
'Cs137': 30.17, # major byproduct of Fukushima meltdowns
'Cs134': 2.06,
'Xe133': 5.2475/365.2181, # fraction of a year
'Co60' : 5.27
}
column = Series([float(v) for v in half_life_table.values()], index=half_life_table.keys())
table = DataFrame({'Years':column}, index=column.index)
table.index.name = "Isotope"
table.sort_values(by='Years', inplace=True, ascending=False)
ax = table.plot.bar(logy=True, legend=False, title="Half Lives")
ax.set_ylabel("Years (log scale)");
The fact that the atomic nucleii of unstable isotopes kick out particles (shoot bullets) in order to become more stable, is only the beginning of the story.
Exactly what particles do they kick out? Neutrons? Alpha particles? Electrons?
The type of particles emitted makes a difference when it comes to the effects on biologicals (see below). Just counting their number, like a Geiger counter does, only tells part of the story.
Although an isolated proton in a hydrogen atom is not known to spontaneously decay, protons within atomic nucleii will turn into neutrons, and vice versa.
When a proton turns into a neutron, it ejects a positron and a neutrino (β+ decay). When a neutron turns into a proton, it kicks out an electron and an anti-neutrino (β- decay). See Fig. 620.06 in Synergetics.
A proton may capture an electron from the surroundings and turn into a neutron, emiting a neutrino.

Permission details:
CC-BY-SA-3.0-MIGRATED; Licensed under the GFDL by the author; Released under the GNU Free Documentation License.
YouTubeVideo("0EBTn_3DBYo") # https://youtu.be/0EBTn_3DBYo
Historical Sidebar¶
New Zealand based geochemist Bernie Gunn used to complain to me (Kirby Urner) that the new generations of geochemist seemed mostly eager to attend conferences at resort destinations and enjoy drinks by the pool. When would they find the time to get any work done?
Programming Interlude: Ploting Radioactive Decay¶
α Decay:
21084Po⟶42He+20682Pbor21084Po⟶42α+20682Pbβ+ Decay:
4019K+0−1e⟶4018Ar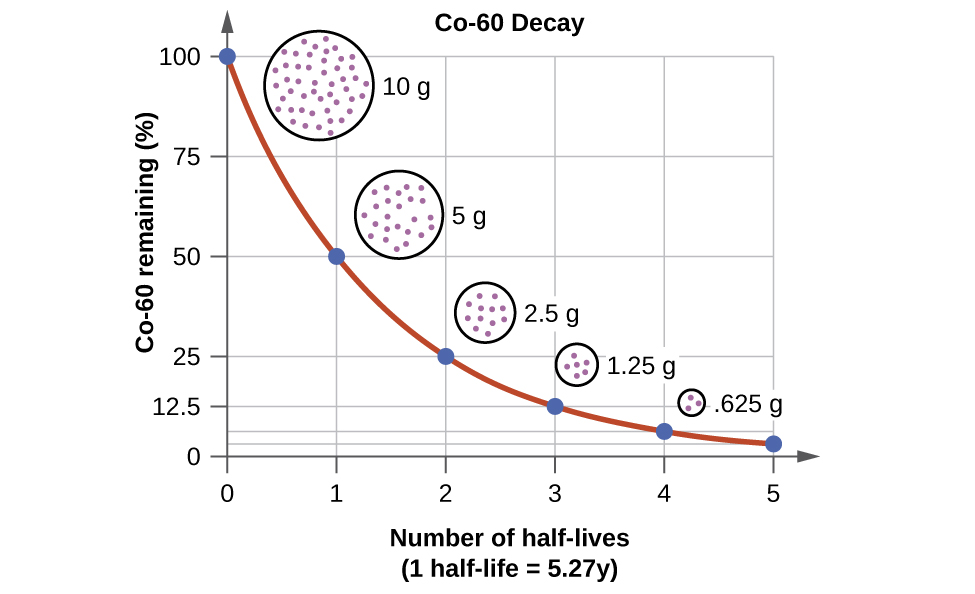
For more background on plotting using matplotlib see Arithmetic, Plotting.
%matplotlib inline
import numpy as np
import pandas as pd
import matplotlib.pyplot as plt
import math
half_life_colbalt = 5.27 # years
rate_constant = math.log(2)/half_life_colbalt
rate_constant
0.13152697923338622
time_axis = np.arange(0, 6 * half_life_colbalt, half_life_colbalt/12) # 100 data points
snap_shots = np.linspace(0, 6 * half_life_colbalt, 6)
remaining_cobolt_cont = 100 * math.e ** (-rate_constant * time_axis)
remaining_cobolt_disc = 100 * math.e ** (-rate_constant * snap_shots)
year15 = 100 * math.e ** (-rate_constant * 15)
year15
13.905236523299653
# high frequency sample (more continuous)
data1 = pd.DataFrame({"Time": pd.Series(time_axis),
"Mass": pd.Series(remaining_cobolt_cont)})
# low frequency sample (more discrete)
data2 = pd.DataFrame({"Time": pd.Series(snap_shots),
"Mass": pd.Series(remaining_cobolt_disc)})
data1.head()
| Time | Mass | |
|---|---|---|
| 0 | 0.000000 | 100.000000 |
| 1 | 0.439167 | 94.387431 |
| 2 | 0.878333 | 89.089872 |
| 3 | 1.317500 | 84.089642 |
| 4 | 1.756667 | 79.370053 |
data2.head(6)
| Time | Mass | |
|---|---|---|
| 0 | 0.000 | 100.000000 |
| 1 | 6.324 | 43.527528 |
| 2 | 12.648 | 18.946457 |
| 3 | 18.972 | 8.246924 |
| 4 | 25.296 | 3.589682 |
| 5 | 31.620 | 1.562500 |
Tips:
The DataFrame.plot method does a lot. You may set the kind of plot, and in extracting the axes, you've got access to an Artist.
Getting multiple plots in the same figure requires passing the axes into subsequent plots. StackOverFlow has the answers a lot of times.
ax = data1.plot(x="Time", y="Mass", kind="line", color = ['c'])
ax.set_ylim(0,105)
# note passing ax
ax = data2.plot.scatter(x="Time" ,y="Mass", ax = ax,
color = 'tab:orange', s=40)
ax.set_xlabel("Years")
ax.set_ylabel("Cobalt Remaining")
/Users/kirbyurner/opt/anaconda3/lib/python3.9/site-packages/pandas/plotting/_matplotlib/core.py:1114: UserWarning: No data for colormapping provided via 'c'. Parameters 'cmap' will be ignored scatter = ax.scatter(
Text(0, 0.5, 'Cobalt Remaining')
Plutonium is still omnipresent in the environment from previous atmospheric nuclear weapons tests. We investigated soil and plants sampled at different hot spots in Japan, searching for reactor-borne plutonium using its isotopic ratio 240Pu/239Pu. By using accelerator mass spectrometry, we clearly demonstrated the release of Pu from the Fukushima Daiichi power plant: While most samples contained only the radionuclide signature of fallout plutonium, there is at least one vegetation sample whose isotope ratio (0.381 +/- 0.046) evidences that the Pu originates from a nuclear reactor (239+240Pu activity concentration 0.49 Bq/kg). Plutonium content and isotope ratios differ considerably even for very close sampling locations, e.g. the soil and the plants growing on it. This strong localization indicates a particulate Pu release, which is of high radiological risk if incorporated.

License
21.3 Radioactive Decay by Rice University is licensed under a Creative Commons Attribution 4.0 International License, except where otherwise noted.
Useful Units:
- Joule "In electrical terms, the joule equals one watt-second— i.e., the energy released in one second by a current of one ampere through a resistance of one ohm."
- Becquerel (Bq) = one radioactive decay event per second.
- The Gray (Gy) = one joule per kilo of radioactivity
- The Rad: 1/100th of a gray (same as rem)
- Banana Equivalent Dose (BED)
- The sievert replaces a former unit, the rem : 1 Sv = 100 rem. The Sv and rem take into account the toxicity of the radiation through use of weighting factors. Is it made up of neutrons, alpha particles, gamma rays, or what? What organs were exposed?
The energy units such as sievert are not power units (Energy per Time). Regulatory standards are often expressed in terms of milli-sieverts per year. 1 mSv / yr per person is considered safe but then actual annual exposure is usually higher. Nuclear plant workers get about 50 mSv / yr,
Equivalent Dose (H) = Grays * WR (WR = type of radiation)
For x-rays and gamma rays, 10 milligray (mGy) = 10 mSv For neutrons, 10 milligray (mGy) = 50-200 mSv For alpha radiation (helium-4 nuclei), 10 milligray (mGy) = 200 mSv
WR = pd.Series([1,1,20], index=['Gamma',"Beta", 'Alpha'])
WR
Gamma 1 Beta 1 Alpha 20 dtype: int64
Effective Dose (E) = Grays * WR * WT (WT = tissue weighting factor)
body_parts = [
"Bladder",
"Bone marrow (red)",
"Bone surface",
"Brain",
"Breast",
"Colon",
"Esophagus",
"Gonads",
"Liver",
"Lung",
"Salivary glands",
"Skin",
"Stomach",
"Thyroid",
"Remainder"]
ICRP103 = [0.04, 0.12, 0.01, 0.01, 0.12, 0.12, 0.04, 0.08, 0.04, 0.12, 0.01, 0.01, 0.12, 0.04, 0.12]
WT = pd.Series(ICRP103, index=body_parts)
WT
Bladder 0.04 Bone marrow (red) 0.12 Bone surface 0.01 Brain 0.01 Breast 0.12 Colon 0.12 Esophagus 0.04 Gonads 0.08 Liver 0.04 Lung 0.12 Salivary glands 0.01 Skin 0.01 Stomach 0.12 Thyroid 0.04 Remainder 0.12 dtype: float64
Canned colormaps may be sampled and cycled through.
cs = plt.cm.tab20(np.linspace(0, 1, 15)) # snip a canned strip into colored pieces
ax = WT.plot.pie(figsize=(6,6), labels=None, colors=cs)
ax.set_ylabel('')
ax.set_title("Tissue Weighting (ICRP103)", fontsize=20)
plt.legend(bbox_to_anchor=(.1,.5), labels=WT.index, loc="right");
"[O]ne sievert is generally defined as the amount of radiation roughly equivalent in biological effectiveness to one gray (or 100 rads) of gamma radiation." Britannica
1 mSv is the dose produced by exposure to 1 milligray (mGy) of radiation.

Clearly the sievert and rem depend on a lot of ancillary science regarding the adverse health effects of different types of radiation and from what distance. There's room to cook the books, such as by excluding certain radio frequencies from the ionizing category.
Electromagnetic radiation is a source of sieverts in the case of gamma rays and microwaves. If you stand close to the wrong frequencies of electromagnetic radiation (high frequency photons), you could get burned.
So what about 5G, the new cell phone frequencies, held close to the head or transmitted from a nearby light pole? "More study is needed" say some. "No significant increased risk" say others. Be on the lookout for junk science wherever politics are involved.


Will Asylum City have a 5G Quarter?¶
YouTubeVideo("YVSmf_qmkbg")
YouTubeVideo("SuQI_g__BaU")
YouTubeVideo("nHDaRPQ8aiM")
YouTubeVideo("iUE2846zgCA") # https://youtu.be/iUE2846zgCA
YouTubeVideo("JKaoLxw0qJI") # https://youtu.be/JKaoLxw0qJI
YouTubeVideo("Cc-vvhWXL9Q") # https://youtu.be/Cc-vvhWXL9Q
YouTubeVideo("dur-q-r0pW4") # https://youtu.be/dur-q-r0pW4
YouTubeVideo("NzqJtHZXsiM") # https://youtu.be/NzqJtHZXsiM
YouTubeVideo("J-hfoDy904E") # https://youtu.be/J-hfoDy904E
YouTubeVideo("GXefbSeo9HM") # https://youtu.be/GXefbSeo9HM
YouTubeVideo("WFCvkkDSfIU") # https://youtu.be/WFCvkkDSfIU
YouTubeVideo("A83aetwlOSc") # https://youtu.be/A83aetwlOSc
YouTubeVideo("_aL_hY8DjMU") # https://www.youtube.com/live/_aL_hY8DjMU